Blog
Sodium-ion Batteries Explained: A Sustainable Alternative to Lithium-ion?
Summary: Sodium-ion batteries (SIBs) use abundant sodium instead of scarce lithium.
✅ 30–40% cheaper • ✅ Works at -40°C • ✅ Non-flammable
🚧 Lower energy density (150 Wh/kg) • 🚧 Shorter cycle life
Best for: grid storage, e-bikes, cold climates
First mass-produced SIB EV: JAC Yiwei 3 (2024)
Prediction: 20–30% of stationary storage by 2030
Scroll for the full story →
What is a Sodium-Ion Battery (SIB)?
Lithium-ion batteries power nearly every aspect of our modern life — from smartphones and industrial automation to energy storage systems and electric mobility. They have become the dominant energy source in our daily lives.
However, as global electrification accelerates, the growing demand for lithium and other key materials such as cobalt and nickel has led to rising costs, resource scarcity, and environmental concerns associated with mining and battery disposal.
To address these challenges, sodium-ion battery (SIB) technology has emerged as a promising alternative. Sodium-ion batteries are rechargeable batteries that rely on the movement of sodium ions between the positive and negative electrodes to complete charging and discharging.
With their abundant raw materials, lower cost, improved safety, wider temperature tolerance, and reduced reliance on lithium, cobalt, and nickel, sodium-ion batteries are gaining attention in both stationary energy storage and electric mobility applications.

How Does a Sodium-Ion Battery Work?
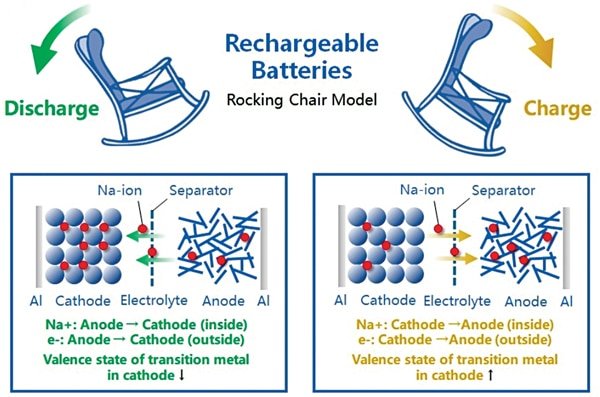
Sodium-ion batteries are often called “rocking-chair batteries” because their working principle is similar to that of lithium-ion batteries.
A sodium-ion battery cell consists of a cathode, anode, separator, electrolyte, and current collectors. The separator isolates the cathode and anode to prevent short circuits, while the electrolyte allows sodium ions (Na⁺) to move between them.
During charging, sodium ions are extracted from the cathode, move through the electrolyte, and enter the anode, creating a sodium-rich anode and a sodium-poor cathode. Electrons flow through an external circuit to maintain charge balance.
During discharging, the process reverses — sodium ions move back to the cathode while electrons flow through the circuit to provide power.
Because both processes are symmetrical and rely on sodium ion movement, sodium-ion batteries operate on the same fundamental principle as lithium-ion batteries.
Advantages of Sodium-Ion Batteries
- Cost Advantage
Sodium is the sixth most abundant element in the Earth’s crust (2.75%) and is evenly distributed globally. In contrast, lithium (0.0065%) is scarce and concentrated mainly in South America.
SIBs can also use aluminum for both current collectors, further reducing material costs. In current production setups, sodium-ion batteries can achieve 30–40% cost savings compared to lithium iron phosphate (LFP) batteries. - Excellent Low-Temperature Performance
In low-temperature tests, sodium-ion batteries (copper-based oxide/carbon system) retain over 88% capacity at -20°C, while lithium-ion batteries (LFP/graphite system) maintain less than 70%. - High Charging and Discharging Rate
Sodium ions have lower solvation energy and a smaller Stokes diameter than lithium ions, resulting in faster interfacial ion diffusion and higher ionic conductivity. This allows for faster charging under similar conditions. - High Safety Performance
Sodium-ion batteries are inherently non-flammable, partly due to higher internal resistance and stable chemistry. This gives them an advantage in safety-critical applications.
Challenges and Limitations
The main drawback of sodium-ion batteries lies in the nature of sodium ions — they are about 25% larger than lithium ions. This reduces ion mobility, slows electrochemical reactions, and results in lower energy density (around 150 Wh/kg at the cell level) compared to 180–300 Wh/kg for lithium-ion batteries.
Cycle life is also shorter: 2,000–3,000 cycles for sodium-ion batteries versus 5,000–10,000 cycles for modern LFP cells.
However, rapid advancements in materials and engineering are steadily narrowing these gaps.
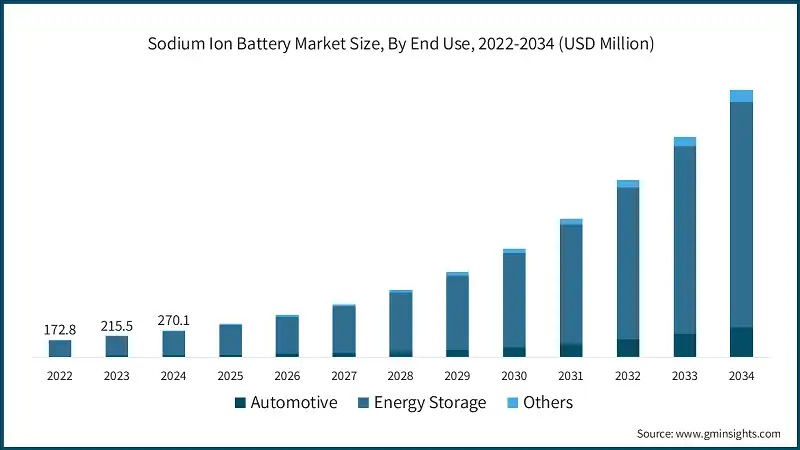
Current Applications and Market Landscape
Sodium-ion batteries are now transitioning from R&D to commercial validation. Rather than replacing lithium-ion batteries, they are expected to complement them, serving different application scenarios based on cost, performance, and temperature resilience.
They are especially well-suited for:
- Stationary energy storage systems
- Commercial and industrial equipment
- Heavy-duty machinery
- Low-speed electric mobility (e-bikes, delivery vehicles, etc.)
In hybrid systems, sodium-ion batteries can also work alongside fuel engines to enhance performance, reduce fuel consumption, and extend system life.
Future Prospects and Innovations
With continued research and industrial investment, sodium-ion batteries are expected to play a growing role in the global energy mix. Their low cost, material abundance, and strong safety make them ideal for large-scale energy storage and emerging electric mobility markets.
Conclusion
Sodium-ion batteries represent a practical and sustainable pathway toward large-scale electrification. While they won’t replace lithium-ion batteries entirely, their complementary strengths — affordability, safety, and cold-weather resilience — make them a vital part of the next-generation energy landscape.
